|
Ī vaccine being developed by Maryland-based Novavax is using an ethically-derived invertebrate cell line Sf9 to produce protein nanoparticle antigens that make its vaccine work. U.K.-based GSK and France-based Sanofi are the world’s #1 and #3 largest vaccine producers respectively by total revenue in 2017 according to FiercePharma. Sanofi Pasteur will be bringing to the table an ethically produced antigen for the vaccine and GSK will be contributing an adjuvant-an immune response booster that improves the effectiveness of a vaccine. Of particular note, rival pharmaceutical giants Sanofi Pasteur and GSK have teamed up in an unprecedented partnership to jointly develop a vaccine for SARS-CoV2. While many COVID-19 vaccines are being developed with fetal cell lines, a number of promising vaccine candidates, such as those being developed by Novavax, Sanofi Pasteur, GlaxoSmithKline (GSK), and Sinovac, are using ethically-derived cell lines. Anthony Fauci.Įthically-Produced COVID-19 Vaccines in the Pipeline NIAID is a division of the National Institutes of Health (NIH) led by Dr. Moderna is also receiving substantial research assistance for its COVID-19 vaccine from the National Institute of Allergy and Infectious Diseases (NIAID) which helped develop the vaccine and conduct clinical trials. 
Centers of Disease Control and Prevention (CDC) has been producing samples of the SARS-CoV2 virus for biotech and pharmaceutical companies to use for vaccine research using fetal HEK-293T cells (a decedent cell line of HEK-293). BARDA’s funding for Emergent, however, was not awarded specifically for scaling up production of J&J’s vaccine candidate. Emergent BioSolutions is currently working with Janssen of Johnson & Johnson to manufacture their trial vaccines. to accelerate development and manufacturing capacity for COVID-19 vaccines and drug treatments. On June 1st, BARDA issued a $628 million task order under a preexisting government contract with Emergent BioSolutions Inc. Inovio has also received an unspecified grant for developing its vaccine candidate from the Defense Advanced Research Projects Agency (DARPA) at the Department of Defense. BARDA has also made grants for up to $483 million for Moderna’s vaccine and $456 million for Janssen Research and Development, LLC of Johnson & Johnson. Department of Health and Human Services (HHS).īARDA has awarded a $1.2 billion grant for AstraZeneca to fund research for the trial vaccine it is jointly developing with Oxford University. Most of this funding has been awarded through the Biomedical Advanced Research and Development Authority (BARDA), a division within the U.S. government has made grants totaling nearly $2 billion in support of the development of COVID-19 vaccines using fetal cell lines. The PER.C6 fetal cell line was derived from retinal tissue taken from an 18-week-old baby boy who was aborted in the Netherlands in 1985 and later converted into a fetal cell line in 1995. HEK-293 was originally derived from kidney tissue taken from a baby girl who was aborted in the Netherlands in 1972 and later developed into a cell line in a lab in 1973.Īdditionally, Janssen, the pharmaceutical division of consumer product giant Johnson & Johnson, is using the human fetal cell line PER.C6 to develop its vaccine. Several COVID-19 vaccine frontrunners, including those being developed by Moderna, Oxford University/AstraZeneca, CanSino Biologics/Beijing Institute of Biotechnology, and Inovio Pharmaceuticals, are using a human fetal kidney cell line called HEK-293 to develop their trial vaccines. Several more candidates are expected to begin clinical trials before the end of the year. Of these, 10 vaccine candidates have already advanced to clinical trials to test the vaccine candidate’s safety and efficacy. Pharmaceutical companies are sprinting to have a vaccine ready by the end of the year or by early 2021.Īccording to a tracker from the World Health Organization, there are now more than 120 vaccine candidates in development. From big pharma to small biotech companies and universities, researchers have been pushing out dozens of vaccine candidates and have fast-tracked promising vaccine candidates to clinical trials in record time. With more than 6.2 million reported cases so far and more than 375,000 deaths worldwide, the burden of disease from the 2019 novel coronavirus continues to mount. 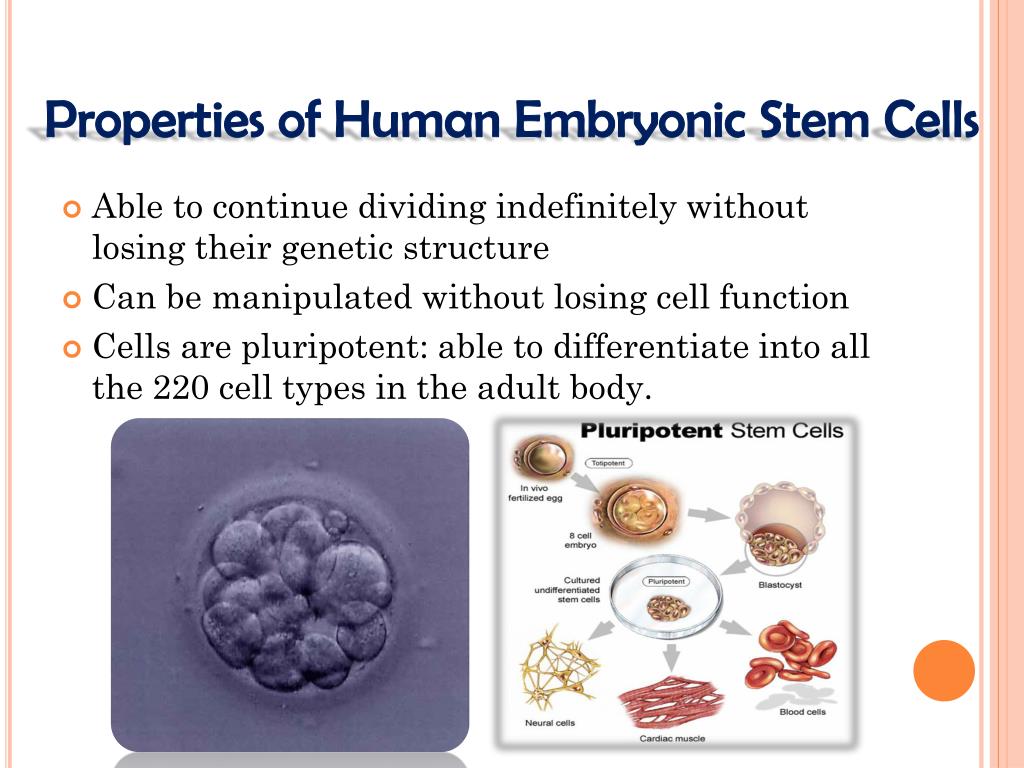
The bad news is that many of the leading vaccine candidates for the 2019 novel coronavirus (SARS-CoV2) are being developed using fetal cell lines that were originally derived from the tissues of aborted babies in the 1970s and 80s. The good news is that many of the world’s largest vaccine companies are developing promising vaccine candidates using ethically-derived cells. The race is on to find a vaccine for COVID-19.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
